On the weekend, Dr. Schett specially invited Popeye, Olive Oyl, and Bluto to visit his lab, letting them see firsthand how immune tolerance induction therapy is developed and feel the “scientific magic” in the lab. Walking into the lab, a faint smell of disinfectant greeted them. All kinds of precision instruments were lined up neatly, glowing softly. Scientists in white coats were operating the equipment attentively, their expressions serious and rigorous.
Bluto leaned in curiously, staring at a running centrifuge, and asked quietly: “Dr. Schett, what’s this instrument for? It looks so amazing—like magic.” Dr. Schett walked over with a smile, patting Bluto’s shoulder: “Young man, this isn’t magic—it’s the power of science. This centrifuge is used to extract and screen self-antigens. It’s the first and most basic step in developing immune tolerance induction therapy.”

“Extracting and screening self-antigens?” Olive Oyl asked, curious. “Dr. Schett, how exactly is this done?” Dr. Schett took them to another instrument, pointing to the sample inside: “Look. These samples are all taken from patients with autoimmune diseases, containing various cells and antigens. Our first step is to extract the self-antigens that may be misjudged by the immune system from these samples—like joint synovial antigens and nerve myelin antigens.”
He paused, continuing: “Just like Popeye picks the best spinach to get enough strength, we also need to pick the purest antigens that can induce tolerance accurately, eliminate useless components, and avoid causing abnormal immune reactions. This step requires precision instruments, repeated screening and purification, to ensure that the extracted antigens can be recognized by the immune system accurately, helping the immune patrol recognize their own people again.”
“Can the antigens be used directly for treatment after extraction?” Bluto asked. Dr. Schett shook his head: “Of course not. After extracting high-quality antigens, we need to take the second step—preparation and optimization of antigen preparations. Just like Popeye doesn’t eat spinach powder directly, we need to process it. We also need to combine the antigens with carriers like liposomes and nanoparticles to make low-dose preparations.”
Popeye added: “These carriers are like a ‘protective shield,’ keeping the antigens from being digested by stomach acid and reaching the immune system’s ‘training base’ smoothly. At the same time, we need to optimize the concentration and delivery efficiency of the preparation, ensuring that the antigens can reach the ‘trainers’ like dendritic cells and T cells safely and accurately—ensuring the induction effect while reducing side effects. This step requires repeated experiments and formula adjustments. Just like I practice rowing repeatedly to master the best skills, scientists also need to try repeatedly to develop the most suitable preparation.”
“What’s the third step?” Olive Oyl asked. Dr. Schett smiled: “The third step is animal experiments and clinical research. This is the key to verifying the treatment effect and safety, and also the longest and most rigorous step. We first test it on experimental animals like mice and monkeys, observe the animals’ immune reactions, symptom relief, and evaluate the effectiveness and safety of the treatment. For example, in mouse models of multiple sclerosis, we test whether the antigen preparation can activate Treg cells, suppress ‘rebellious’ cells, and relieve the mice’s symptoms like limb numbness and mobility impairment.”
He continued: “Only when animal experiments achieve significant results can we enter the clinical research stage. In the clinical research stage, we recruit patients with autoimmune diseases, divide them into treatment groups and control groups. The treatment group uses immune tolerance induction preparations, and the control group uses placebos or traditional drugs. Through long-term observation, we compare the symptom relief, recurrence rate, and side effect rate of the two groups of patients, further optimizing the treatment plan. This process often takes several years—just like Popeye insists on eating spinach and exercising to stay strong, the R&D process also requires patience and persistence to ensure the safety and effectiveness of the treatment.”
Bluto looked at the busy scientists in the lab and couldn’t help sighing, “I had no idea so much hard work went into developing this treatment. Scientists really have it tough!”
Popeye nodded in agreement. “Every scientific achievement comes from repeated trials and perseverance. Just like how I protect the harbor by exercising every day and staying alert against danger, scientists keep working hard to help more people break free from the pain of illness.”
Dr. Schett added, “Our research and development is still progressing. We hope to further shorten the treatment cycle and expand its scope of application through more precise preparation techniques. In the next episode, we will hear real treatment stories from patients with autoimmune diseases, and see how immune tolerance induction therapy has changed their lives and brought them hope.”
Science behind
Process of Drug Development Process
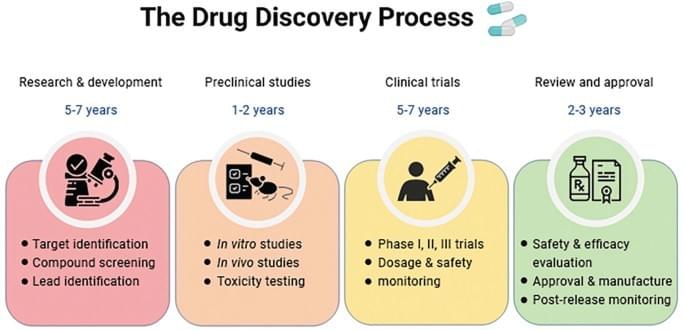
First is the drug discovery stage, which takes 5 to 6 years excluding preliminary preparation work. Researchers screen candidate drugs from tens of thousands of potentially effective substances. At the final step of this stage (preclinical research), further screening is conducted through in vitro experiments (test tube experiments) and in vivo experiments (animal experiments). On average, only 1 out of 25 selected potential substances can enter the next round of testing. Overall, only 10 out of 10,000 candidate substances are eligible for clinical trials.
Next is the clinical trial stage, which takes 5 to 7 years and is carried out in three phases step by step: Phase 1 involves 20 to 100 healthy volunteers, mainly testing the safety of the drug and the appropriate dosage; Phase 2 recruits hundreds of patients with the disease, focusing on whether the drug is effective and whether it has side effects; Phase 3 is the longest, with up to thousands of patients participating to further verify the efficacy and safety. Even if a drug can enter clinical trials, less than 10% of them can successfully complete all three phases and obtain approval from official drug regulatory authorities.
The drug approval stage involves experts from various fields such as doctors, chemists, and microbiologists conducting joint evaluations. The core is to determine whether the benefits of the new drug outweigh its potential risks. Only if it meets the requirements can it obtain marketing approval, but this is not the end of the process.
Finally, there is post-market safety monitoring. After the drug is launched, its usage data will be continuously collected. If the regulatory authorities find problems with the drug, they will require revising the dosage, adding usage warnings; if the problems are relatively serious, they will even directly remove the drug from the market to ensure the safety of patients' medication.
In simple terms, the birth of a new drug requires "layer-by-layer screening, strict testing, and full-cycle monitoring". It takes a long time and has high thresholds, and every step is to ensure the safety and effectiveness of medication.

